
Alginate Structure
Alginate is an anionic natural polymer that is composed of glucuronic acids (linear block copolymers). The glucuronic acids that make up alginate are β-D-mannuronic acid (M) and α-L-guluronic acid (G)6.

The M acids are able to form β-1,4-linkages, these links give the M blocks a flexible and linear confirmation. Whilst the G acids form α-1,4-linkages, these links provides the polymer with a folded and rigid structure (due to steric hindrance of the carbonyl group).
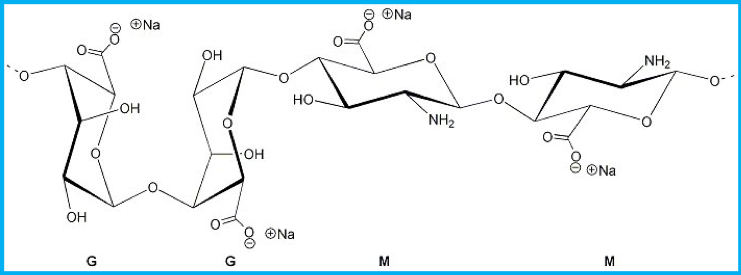
Figure 3 Formation of M-blocks, G-blocks and MG-blocks. This image was obtained from Wikimedia commons
Alginate gels are 3D cross-linked networks, there are many methods in order to achieve this cross linking such as thermal gelation, cell cross-linking, covalent cross-linking and ionic covalent. I’ll be focusing on the ionic cross-linking as it is the most common method to prepare these alginate gels, it involves combining an alginate solution with divalent cations such as Ca2+. The divalent cations will bind to the G-blocks which allows them to form a junction with other G-blocks from adjacent chains, this forms the gel structure6. There are however disadvantages in this method, the most critical disadvantage is that there is a limited long-term stability in some physiological conditions, as the gel will dissolve when the divalent cations are released into the surrounding media due to the exchange reactions with monovalent cations6.